If you've been pricing peptides online and noticed wildly different prices for the same molecule, the honest answer is: the cheap source is the illegal one, the legal path looks more like a pharmacy than a marketplace, and skipping the prescriber to save money is how people end up with contaminated injections. A 10 mg vial of a research-grade peptide from an online vendor might cost $30. The same active molecule dispensed by a licensed 503A compounding pharmacy under a physician's prescription costs considerably more, because it comes with manufacturing oversight, a Certificate of Analysis, and a pharmacist who can call your prescriber if something looks wrong. The price difference is not a market inefficiency you can exploit. It reflects the presence or absence of the entire quality and safety system built around that product. Understanding how that system is structured is the clearest way to protect yourself before you make any decision about peptides.
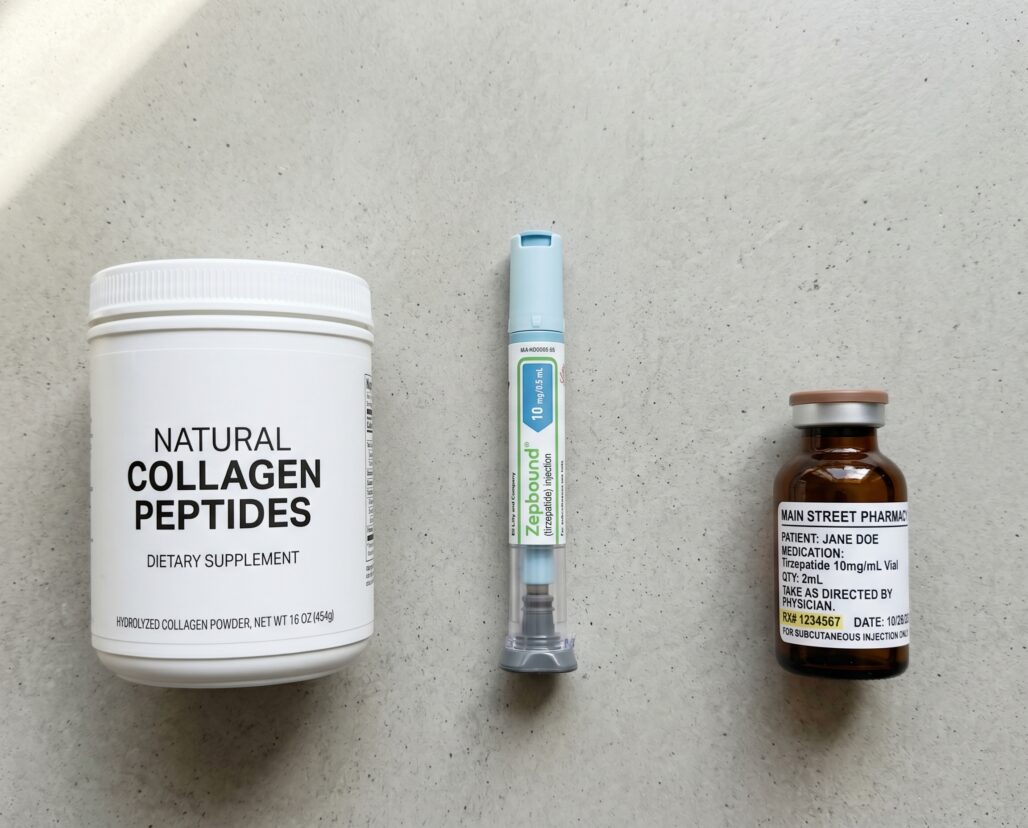
Summary: Three Legal Tiers, One Illegal Category
The US market for peptide products splits cleanly into three legitimate tiers and one category that sits outside the law regardless of how it is labeled.
Tier 1 covers consumer products that contain peptide ingredients and are sold legally over the counter: collagen peptide powders, copper-peptide cosmetic serums, and similar products regulated as dietary supplements or cosmetics. These require no prescription and carry no meaningful regulatory barrier to purchase.
Tier 2 covers FDA-approved peptide drugs. These are prescription medications reviewed for safety and efficacy by the FDA, dispensed only by licensed pharmacies, and available only to patients with a valid prescription from a licensed prescriber.
Tier 3 covers compounded peptide drugs prepared by licensed 503A or 503B pharmacies. These are a regulated middle ground: not FDA-approved in the same way as branded drugs, but subject to legal manufacturing standards and requiring a prescription from a licensed healthcare provider.
The illegal category covers websites selling injectable peptides labeled "for research use only" or "not for human consumption." The FDA has been explicit: this label does not create a lawful consumer pathway. These products are unapproved drugs.
| Tier | Where | Examples | Legal Status | Safety Profile |
|---|---|---|---|---|
| 1 — OTC Consumer | Supplement retailers, beauty stores | Collagen peptide powders, GHK-Cu serums | Legal, no Rx needed | Recognized safe within category; limited clinical depth |
| 2 — FDA-Approved Rx | Licensed pharmacy with valid Rx | Semaglutide (Ozempic/Wegovy), tesamorelin (Egrifta) | Fully legal with prescription | FDA-reviewed; prescribing information filed publicly |
| 3 — Compounded Rx | 503A/503B licensed pharmacy with valid Rx | Compounded sermorelin, compounded GLP-1 formulations | Legal with Rx; subject to compounding law | Varies by pharmacy; 503B under stricter manufacturing standards |
| Grey Market | Online research-chemical vendors | Injectable BPC-157, unlabeled semaglutide salts | Not a legal consumer pathway; FDA warning letters issued | Unknown; impurity levels up to 24% documented in analyses |
Tier 1: OTC Consumer Products With Peptide Ingredients
The easiest legal path is a supplement store. Collagen peptide powders are among the best-selling protein supplements in the country. GHK-Cu (copper peptide) appears in dozens of cosmetic serums applied topically rather than injected.
These products occupy a different regulatory space than injectable peptide drugs. Collagen hydrolysate sold as a dietary supplement is regulated under the Dietary Supplement Health and Education Act of 1994, which does not require pre-market FDA approval. Copper-peptide cosmetics are regulated as cosmetics under the FD&C Act. Neither requires a prescription, and neither carries the risk profile of an injectable drug that bypasses the skin and gut entirely.
The practical rule for Tier 1: if a peptide product is sold in a supplement or beauty retailer without a prescription, it is almost certainly a topical or ingestible product regulated under supplement or cosmetic law. No legitimate injectable peptide is sold legally without a prescription.
Tier 2: FDA-Approved Peptide Drugs From Licensed Prescribers
Several peptide drugs have gone through the full FDA new drug application process and carry approved prescribing information — known dosing, known side-effect profiles, and manufacturing standards enforced through FDA inspections. The article FDA-Approved Peptides on this site covers the full approved list in detail.
For these drugs, the legal path is uncomplicated: a licensed prescriber evaluates your situation, writes a prescription, and a licensed pharmacy dispenses the drug. You receive a product with a full chain of custody from FDA-registered manufacturer to pharmacy to your hands.
The friction is cost, not legality. Branded GLP-1 agonists cost several hundred dollars per month without insurance. These costs push some patients toward compounding pharmacies (Tier 3) or grey-market sources. Understanding the distinction between those two alternatives is the most practically important thing in this article.
Tier 3: 503A and 503B Compounding Pharmacies
Compounding pharmacies occupy a specific legal space created by the Drug Quality and Security Act of 2013, which established two distinct tracks under Section 503A and Section 503B of the Federal Food, Drug, and Cosmetic Act.
503A pharmacies are traditional compounding pharmacies: state-licensed, filling individual patient-specific prescriptions. A 503A pharmacy compounds a drug for a named patient under a prescription from a licensed prescriber. These pharmacies are primarily regulated by their state pharmacy boards, though the FDA retains oversight authority and can take action if safety concerns arise. A 503A pharmacy may compound using bulk drug substances, but only those that appear on the FDA's 503A Bulks List or that meet other statutory criteria. If a substance does not appear on that list and is not a component of an FDA-approved drug, a 503A pharmacy cannot lawfully include it in a compounded preparation.
503B outsourcing facilities operate under stricter federal oversight. They can produce drugs in larger batches without individual patient prescriptions, but they must comply with current Good Manufacturing Practice (cGMP) standards, the same manufacturing framework applied to commercial pharmaceutical manufacturers. The FDA inspects 503B facilities on a regular schedule. A 503B facility may only compound a drug that includes a bulk drug substance if that substance appears on the FDA's 503B Bulks List or on the FDA's current drug shortage list.
The practical difference between 503A and 503B for a patient: a 503A prescription is filled specifically for you, the named individual, by a state-licensed pharmacist. A 503B product may be dispensed from a larger pre-made batch, but the manufacturing standards are substantially higher.
What this means for specific peptides: In September 2023, the FDA classified BPC-157 as a Category 2 bulk drug substance, meaning it may present significant safety risks and 503A compounders that include it in preparations could face regulatory action. CJC-1295 acetate is currently proposed to NOT be included on the 503A Bulks List. Many of the popular performance peptides discussed on wellness forums are simply not on any approved bulk drug substances list, which means no licensed 503A or 503B pharmacy can lawfully compound them, full stop.
The actionable path for Tier 3: find a physician who evaluates your clinical situation, writes a prescription for a peptide that is actually on the appropriate FDA bulks list or shortage list, and directs you to a licensed and accredited compounding pharmacy. The Pharmacy Compounding Accreditation Board (PCAB) accreditation is a reasonable quality signal for 503A pharmacies. For 503B, check whether the facility is registered with the FDA as an outsourcing facility.
Why "Research Peptide" Vendors Are Not a Legal Consumer Option
The grey market operates on a simple legal theory: if a product is labeled "for research use only" and "not for human consumption," it is not being sold as a drug and therefore is not subject to the FDA's drug approval requirements. This theory does not work.
The FDA evaluates a product's intended use based on the totality of evidence, not just the label text. If a vendor sells injectable peptide vials with dosing instructions, markets them for their health effects, and operates social media accounts promoting weight loss or recovery outcomes, the FDA reads that context and classifies the product as an unapproved drug regardless of what the label says.
The evidence of enforcement is not hypothetical. In December 2024, the FDA issued a warning letter to Summit Research Peptides after reviewing the company's website and social media presence, which promoted weight loss outcomes for peptide products labeled for research use only. The FDA cited violations of Sections 505(a) and 301(d) of the Federal Food, Drug, and Cosmetic Act — introducing unapproved new drugs into interstate commerce. In March 2026, the FDA issued warning letters to multiple additional vendors selling peptides including retatrutide and tirzepatide under identical "research only" labeling. The Department of Justice prosecuted Tailor Made Compounding LLC for distributing unapproved peptides including BPC-157, resulting in a $1.79 million forfeiture.
Injectable products carry a specific physical risk on top of the regulatory one. When you inject a substance directly into tissue or a vein, you bypass the gastrointestinal tract, the liver's first-pass metabolism, and the skin — several of the body's primary defenses against contaminants. Independent analyses of grey-market injectable peptides have documented impurity levels up to 24%, including formaldehyde derivatives. Vendors selling outside the pharmaceutical supply chain are not required to provide a Certificate of Analysis, and most do not.
The "research only" label is not a legal shield for buyers either. Purchasing an unapproved injectable drug for human use is not a protected activity, regardless of the label's scientific-sounding language.
The Telehealth-Prescribed Compounded Peptide Reality (GLP-1s)
The clearest recent example of compounding law colliding with consumer demand is GLP-1 agonists. When branded semaglutide entered FDA-declared shortage beginning in 2022, 503B outsourcing facilities could lawfully compound it under the shortage-list exemption. A wave of telehealth platforms built businesses around that window.
The FDA's position changed in 2025. The agency determined the semaglutide shortage had been resolved, ending the period of enforcement discretion for 503A compounders. The FDA also flagged a separate problem: some compounders were using semaglutide sodium and semaglutide acetate — salt forms that are chemically different from the active ingredient in the approved drug — and the agency has stated it does not have information confirming those salts share the same pharmacologic properties as Ozempic or Wegovy.
A telehealth prescription alone does not make a compounded peptide lawful. The legitimate pathway requires three independently verifiable conditions: a licensed prescriber conducting a real clinical evaluation, a pharmacy registered as a 503A or 503B facility, and a drug substance on the applicable FDA list or a current shortage list.
For more on the safety profile of GLP-1 peptides and other compounds, see Are Peptides Safe?, and for documented side effects by compound class, see Peptide Side Effects.
A Note on Personal Importation
The FDA's personal importation policy allows enforcement discretion for individuals importing small quantities of medications for personal use. The policy generally applies when the product is for a serious condition with no effective domestic alternative, when there is no commercial promotion to US residents, and when the quantity does not exceed a 90-day supply.
This discretion exists to help people continue treatment they began abroad. It is not a pathway for importing unapproved injectable peptides from offshore research-chemical vendors. Package clearance through customs is not evidence of lawful importation.

Frequently Asked Questions
Can I buy peptides without a prescription? Legally, yes for Tier 1 products (collagen supplements, topical copper-peptide serums). No for injectable peptide drugs, which require a prescription from a licensed prescriber and dispensing by a licensed pharmacy.
Are compounding pharmacy peptides the same as FDA-approved versions? Not exactly. Compounded drugs are not FDA-reviewed for safety and efficacy the way approved drugs are. They can be a legitimate option when the compounded substance is on the appropriate FDA bulks list and the pharmacy is properly licensed, but they carry more variability than branded approved drugs.
Is there a list of peptides that 503A pharmacies can legally compound? Yes. The FDA maintains the 503A Bulks List. Many popular performance peptides are not on this list. Substances classified as Category 2 (presenting significant safety risks) include BPC-157 as of September 2023.
What should I ask a compounding pharmacy before filling a peptide prescription? Ask for the pharmacy's state license number, its PCAB accreditation status or FDA outsourcing facility registration (for 503B), and a Certificate of Analysis from an independent third-party laboratory for the specific batch.
Does the FDA's personal importation policy cover research peptides I order online? No. The personal importation policy is discretionary and applies to specific circumstances involving personal medical treatment, not to commercial purchases of unapproved drugs from research-chemical vendors.
What happens if I buy from a research-peptide vendor and something goes wrong? You have limited recourse. These vendors are not required to maintain adverse event reporting systems or manufacturing records consistent with licensed drug manufacturers. The FDA and DOJ have demonstrated willingness to pursue vendors criminally, not just issue warning letters.
Conclusion
The legal path to peptides in the US is a tiered system, not a marketplace free-for-all. Consumer peptide products (collagen powders, copper-peptide serums) are legal over the counter and carry the lightest regulatory requirements. FDA-approved peptide drugs require a prescriber and a licensed pharmacy and carry the strongest safety documentation. Compounded peptides from 503A or 503B pharmacies occupy a legitimate middle space, provided the substance is on the appropriate FDA bulk substances list and the pharmacy is properly licensed — conditions that many popular performance peptides and several recently popular GLP-1 formulations currently do not meet.
Research-peptide vendors are not a fourth tier. They are a category the FDA has repeatedly and explicitly characterized as selling unapproved drugs, and the warning letters and criminal prosecutions since 2024 document that the agency is actively enforcing that position.
The actionable path is straightforward: find a licensed prescriber, verify the compounding pharmacy's credentials independently, and confirm the peptide is on the applicable FDA bulk substances or shortage list. That process takes longer and costs more than placing an online order. It also produces a product you can trace, a pharmacist you can call, and a paper trail that protects you.
This article is for informational purposes only and does not constitute medical advice. Peptide drugs are prescription medications in the United States. Consult a licensed healthcare provider before using any peptide drug or compounded medication. The regulatory landscape described reflects publicly available FDA guidance and enforcement actions as of the article's publication date; regulatory status can change. Always verify current FDA guidance directly at fda.gov.
