MK-677 shows up in nearly every "peptide" bodybuilding forum thread, stacked alongside CJC-1295 and ipamorelin, praised for boosting growth hormone without injections. The trouble is that calling it a peptide is factually wrong — and the distinction is not pedantic. MK-677 is a small synthetic molecule, the closest thing the bodybuilding world has to an oral growth-hormone secretagogue, and it carries a documented signal for insulin resistance and fluid retention that the marketing consistently skips. It was developed by Merck, tested in elderly populations, and failed in a hip-fracture trial that was stopped early for a congestive heart failure safety signal. It has never received FDA approval. What it has instead is a reputation built on a misclassification, a genuine GH-boosting mechanism, and a prohibited-list entry at WADA. Here is the honest read.
Summary / Quick Answer
MK-677 is a non-peptide, orally active small molecule that stimulates the growth hormone secretagogue receptor (GHS-R1a). It raises GH and IGF-1 levels meaningfully in both young and elderly populations — that part is real and well-documented. What is also real: insulin sensitivity decreases, fluid retention is common, and a Merck-sponsored hip-fracture trial was halted early after a safety signal for congestive heart failure.
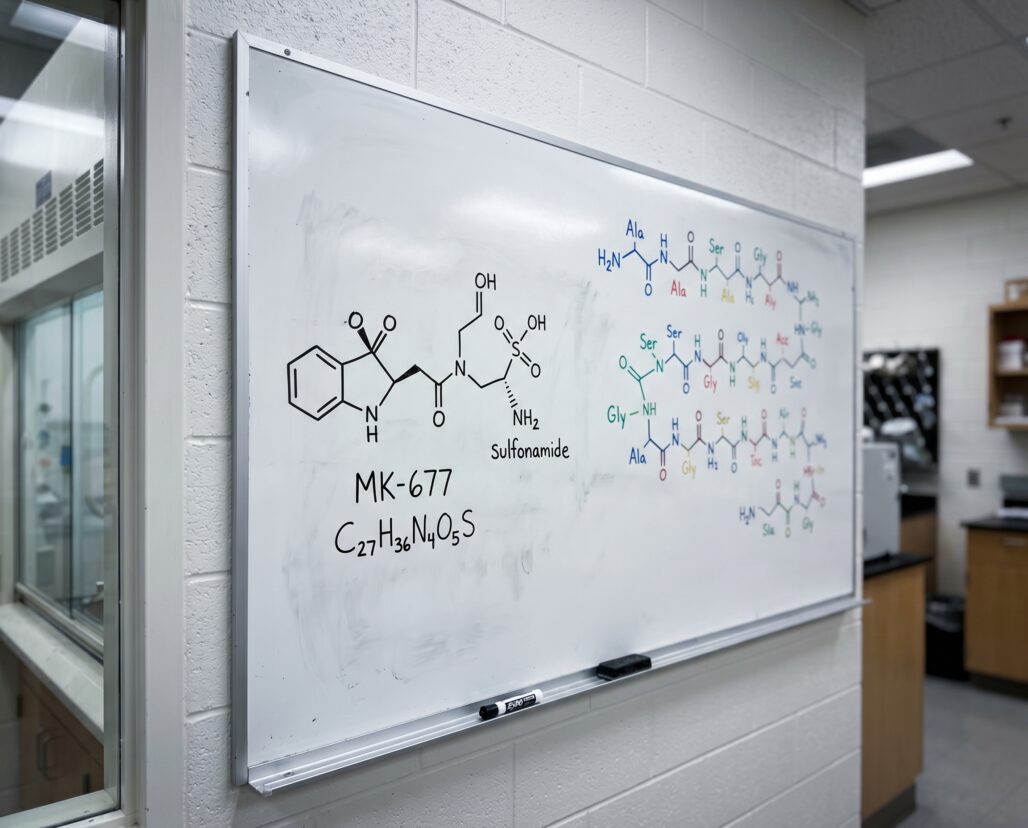
What it is: A heterocyclic small molecule (molecular weight ~528 g/mol) classified as a ghrelin receptor agonist. It is not a peptide — it contains no amino acids, no peptide bonds, and does not share peptide chemistry or pharmacology.
What the trials show: IGF-1 rises to young-adult levels in elderly subjects on 25 mg/day (Nass et al., 2008). Strength and functional outcomes did not improve meaningfully in any published trial.
The safety signals: Fasting glucose increases, insulin sensitivity decreases, lower-extremity edema, increased appetite, weight gain. A phase IIb hip-fracture trial was terminated early due to congestive heart failure events.
Regulatory status: Not FDA-approved. Explicitly named on the 2026 WADA Prohibited List under Section S2.2.4 (Growth Hormone Releasing Factors / GH Secretagogues).
Decision shortcut: If you are a tested athlete, MK-677 use is a doping violation regardless of how it is labeled or sold. If you are not a tested athlete and are considering it for body composition, the evidence shows GH elevation without consistent strength or functional benefit, plus metabolic costs that include measurable insulin resistance.
MK-677 Is NOT a Peptide: The Structural Reality
This distinction deserves its own section because the misclassification is so pervasive that it shapes how people assess the risk.
A peptide is defined by its chemistry: a chain of two or more amino acids linked by peptide bonds (-CO-NH-). Peptides are derived from amino acids, behave like proteins, and are generally broken down by digestive proteases — which is why most peptide drugs require injection rather than oral dosing.
MK-677 is none of that. Its full chemical name is ibutamoren mesylate. Its structure is a complex heterocyclic small molecule built around a spiro-oxindole core — the kind of molecular scaffold a medicinal chemist designs to achieve oral bioavailability and receptor selectivity. It has a molecular weight of approximately 528 g/mol. It contains no amino acids and no peptide bonds. It is structurally closer to a conventional pharmaceutical drug than to anything your body produces as a signaling peptide.
The reason it ends up grouped with peptides in forums and supplement catalogs is functional, not chemical: it works on the same receptor that ghrelin — a 28-amino-acid peptide hormone — activates. Ghrelin is a peptide. MK-677 is a small molecule that mimics ghrelin's effect on the GHS-R1a receptor. That is a relationship similar to a synthetic opioid and an endogenous endorphin: related mechanism, categorically different chemistry.
The chemistry of what peptides actually are matters here because the misclassification changes how people evaluate safety. Peptides tend to degrade quickly in the body and rarely accumulate. Small molecules with oral bioavailability, like MK-677, have entirely different pharmacokinetics, tissue distribution, and off-target potential. A framework built for one does not transfer cleanly to the other.
Why MK-677 Gets Bundled With Peptides
The conflation did not happen by accident. MK-677 entered bodybuilding culture through the same channels as injectable peptide secretagogues like CJC-1295 and GHRP-2 — grey-market research-chemical vendors selling vials and capsules to people pursuing GH elevation without a prescription. Because those compounds and MK-677 all target the GH axis and circulate in the same communities, they got mentally categorized together.
Marketing reinforced it. Selling "growth hormone peptides" is more appealing than "growth hormone secretagogue small molecules," so the former framing won out in product descriptions. By the time anyone looked closely at the chemistry, the category label had calcified.
There is a superficial logic to the grouping: MK-677 mimics ghrelin, and ghrelin is a peptide. But that reasoning would also make beta-blockers a category of catecholamines, which they are not. The receptor target does not determine the chemical class.
The practical consequence is that people assess MK-677 using a peptide risk framework — brief tissue exposure, limited systemic accumulation — when the relevant framework is that of an orally bioavailable pharmaceutical with sustained receptor activity. Those are not the same risk profiles, and the safety questions around peptides used therapeutically do not answer the safety questions around MK-677.
Mechanism: A GHS-R1a Agonist Taken by Mouth
MK-677 activates the growth hormone secretagogue receptor type 1a (GHS-R1a) — the same receptor that ghrelin binds — expressed primarily in the hypothalamus and pituitary. When activated, it triggers the pituitary to release growth hormone in pulses, which in turn stimulates the liver to produce IGF-1.
The oral bioavailability is what set MK-677 apart from injectable peptide GH secretagogues. Merck's chemists engineered the molecule to survive gastric acid and first-pass metabolism — a design feat peptides cannot achieve without structural workarounds. Oral dosing at 25 mg/day maintains 24-hour GH elevation in clinical studies, a different pharmacokinetic profile from the sharper, shorter pulses produced by injectable GHRP compounds.
Because GHS-R1a is expressed across multiple tissues, activation produces effects beyond the pituitary: intense appetite increase (ghrelin is the hunger hormone), changes in cortisol and prolactin, and the peripheral metabolic effects that became the central problem in clinical development. A receptor that evolved to signal hunger does not switch off its other functions just because you want the GH-related downstream effects.
Real Human Trial History
MK-677 has an unusually substantial clinical trial record for a compound that never reached approval — a fact that cuts both ways. It means the evidence is genuine and not just forum anecdote. It also means you can read what the trials actually found, which is more nuanced than the marketing summary.
Svensson et al., 1998 (PMID 9467542) — obese males, 8 weeks. The first major published trial randomized 24 obese men (BMI greater than 30, ages 18-50) to 25 mg MK-677 or placebo daily for eight weeks. IGF-1 rose approximately 40% (p less than 0.001). Fat-free mass increased significantly. Basal metabolic rate increased at two weeks but was not sustained at eight weeks. The metabolic signal that became a theme across all subsequent trials appeared here first: "an oral glucose tolerance test showed impairment of glucose homeostasis at 2 and 8 weeks."
Murphy et al., 2001 (PMID 11238495) — bone density. This Merck-sponsored trial combined MK-677 with alendronate. The combination improved BMD 4.2% versus 2.5% for alendronate alone — a positive signal for the bone application, and confirmation that IGF-1 elevation was reproducible across age groups.
Bach et al., 2004 (PMID 15066065) — elderly hip fracture patients. The first dedicated hip-fracture trial enrolled 161 patients across 13 centers. IGF-1 rose 84% in treated patients. Functional performance measures — the primary endpoints — showed no significant differences between MK-677 and placebo. Raising IGF-1 did not automatically translate into better recovery outcomes in this frail population.
Nass et al., 2008 (PMID 18981485) — healthy older adults, 2-year trial. The most methodologically rigorous long-term MK-677 trial in the published literature. Sixty-five healthy adults aged 60-81 received 25 mg/day in a randomized, double-blind, modified crossover design. IGF-1 rose to levels seen in healthy young adults. Fat-free mass increased 1.1 kg versus a decrease of 0.5 kg in the placebo group (p less than 0.001). Body weight increased 2.7 kg versus 0.8 kg in placebo. Critically: "increased fat-free mass did not result in changes in strength or function." The compound raised body composition numbers without translating into functional improvement.
Sevigny et al., 2008 (PMID 19015485) — Alzheimer's disease, 416 patients. A randomized trial in Neurology found no clinical effect of MK-677 on Alzheimer's progression — the largest completed MK-677 trial by enrollment.
Adunsky et al., 2011 (PMID 21067829) — hip fracture, phase IIb. The most consequential trial for MK-677's development. This multicenter, randomized, placebo-controlled phase IIb study enrolled 123 elderly hip-fracture patients. IGF-1 increased significantly. The trial was terminated early due to a safety signal: congestive heart failure events in a limited number of treated patients. The authors concluded that "MK-677 has an unfavorable safety profile in this patient population." This is the study that effectively ended Merck's development program for MK-677 in the elderly.
Why Development Failed: Insulin Resistance, Fluid Retention, and a Stopped Trial
The Nass 2008 trial documented the metabolic cost clearly. Fasting glucose increased 0.3 mmol/L (approximately 5 mg/dL) in the MK-677 group (p = 0.015), and insulin sensitivity decreased with active treatment. In elderly populations — the target demographic for most of the GH-secretagogue therapeutic rationale — insulin resistance carries real consequences. Type 2 diabetes rates in adults over 65 are substantially higher than in younger populations, and a drug that worsens insulin sensitivity needs a clear efficacy benefit to justify the tradeoff. The functional data did not provide one.
Fluid retention was consistently reported across trials. Nass 2008 documented transient lower-extremity edema. The 2.7 kg total weight gain versus 0.8 kg in placebo included a meaningful water-retention component alongside the lean-mass increase. Ghrelin receptor activation in the kidney and adrenal axis contributes to aldosterone-related fluid retention — a predictable consequence of the mechanism.
The congestive heart failure signal in the Adunsky hip-fracture trial (PMID 21067829) combined that fluid load with an already vulnerable cardiac population. Elderly hip-fracture patients typically carry pre-existing cardiovascular disease, and adding GH-axis stimulation raised a risk that outweighed the functional benefit the trial was designed to demonstrate. Early termination closed the path toward FDA approval for that indication.
The pattern across trials is consistent: MK-677 reliably raises IGF-1 and fat-free mass. It does not reliably improve strength, function, or clinically meaningful outcomes. The metabolic costs — insulin resistance and fluid retention — scale with duration and are most problematic in the populations that GH-axis therapy was intended to help.
For people pursuing muscle growth outcomes, the functional data should give pause. Gaining lean mass on paper while functional strength stays flat is not the same as getting stronger.
Regulatory Status: FDA and WADA
FDA status: MK-677 has never received FDA approval for any indication. It is not an approved drug, not a recognized dietary supplement ingredient, and not available by prescription. Vendors sell it under labels like "for research purposes only" — a workaround that does not change the compound's status under food and drug law. If a substance is marketed for human use, the label language does not exempt it from drug regulations.
WADA status: MK-677 is explicitly named on the 2026 WADA Prohibited List under Section S2.2.4 — Growth Hormone Releasing Factors, specifically "growth hormone secretagogues (GHS) and their mimetics [e.g. anamorelin, capromorelin, ibutamoren (MK-677), ipamorelin, lenomorelin (ghrelin), macimorelin and tabimorelin]." It is prohibited at all times, in competition and out of competition. The prohibition applies to the compound regardless of how it is obtained or labeled.
For competitive athletes subject to anti-doping rules — which in many sports includes recreational competitors, not just professionals — this is a doping violation, not a grey area.

Frequently Asked Questions
Is MK-677 a peptide? No. It is a synthetic small molecule — a heterocyclic ghrelin receptor agonist with a molecular weight of approximately 528 g/mol. It contains no amino acids and no peptide bonds. The word "peptide" on vendor sites is a marketing choice, not a chemical description.
Does MK-677 raise growth hormone? Yes. GH and IGF-1 elevation is one of the most consistent findings across all published trials. The question is what that elevation actually produces in clinical outcomes — and on that measure, the data is substantially less impressive than the mechanism suggests it should be.
What are the main side effects? Increased appetite (often described as intense and sustained for the first weeks of use), fluid retention in the lower extremities, increased fasting glucose, decreased insulin sensitivity, increased cortisol, and weight gain that combines lean mass and water retention. In elderly populations with cardiovascular comorbidities, the fluid load carried a congestive heart failure signal that ended a Merck trial early.
Can it be used for muscle building? The human trial data shows increases in fat-free mass. It does not show corresponding increases in strength or functional performance. Gaining lean mass and gaining functional strength are not the same outcome. For bodybuilding applications, the body-composition effect is real but the strength benefit is not established.
Is MK-677 legal to buy? In most jurisdictions it occupies a regulatory grey zone — not approved as a pharmaceutical, not recognized as a supplement, sold under research-use disclaimers. That grey zone does not mean it is safe, or that the FDA cannot take enforcement action. It is not legal for human therapeutic use in the U.S.
What happens to insulin sensitivity? Ghrelin receptor activation interferes with normal insulin signaling pathways. In the Nass 2008 two-year trial, fasting glucose increased and insulin sensitivity decreased measurably in healthy older adults on 25 mg/day. This is a drug-class effect of GH-axis stimulation, not a compound-specific surprise. Anyone with impaired glucose tolerance or pre-diabetes should treat this signal as a serious concern to discuss with a physician before considering MK-677.
If you are managing a protocol like this, StackMyMed (our companion app) keeps your doses, timing, and interaction checks in one place. It does not replace medical advice — bring the log to your clinician.
Conclusion
MK-677 was developed seriously, tested rigorously, and did not reach approval — not because no one tried, but because the evidence did not support it. The GH and IGF-1 elevation is real. The functional benefit is not, at least not in the populations where trials were run. The metabolic costs — insulin resistance and fluid retention — are real risks, not minor footnotes. And the hip-fracture trial stopped for congestive heart failure is a controlled trial with a DSMB decision behind it, not an anecdote.
If you encounter MK-677 called a peptide, that is inaccurate. If it is described as proven for muscle building or anti-aging, the clinical evidence does not support that. If it is sold under a research-use label, that label does not change its regulatory status or pharmacology. And if you are a tested athlete, it is on the WADA prohibited list by name.
The actionable takeaway: MK-677 is a pharmaceutical compound with pharmaceutical-grade risks. It deserves evaluation as one — not as a supplement, and not as a peptide.
This article is for informational purposes and not medical advice. Peptides, especially those marketed for therapeutic use, can interact with medications and health conditions. Consult a licensed physician before starting any supplement, particularly if you are pregnant, nursing, taking prescription medications, or managing a chronic condition.
